Fake Mounjaro probe expands beyond NCR after seized stock traced to Hyderabad | Gurgaon News
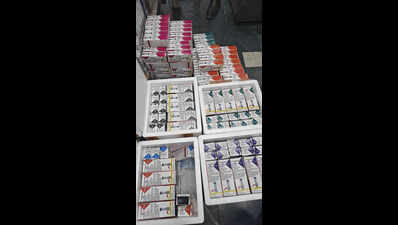
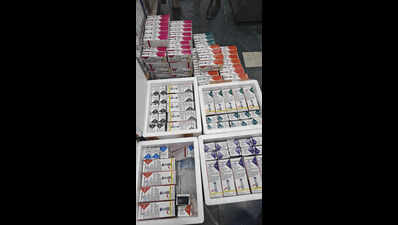
Gurgaon: The drugs control department has widened its probe into distribution of fake Mounjaro injections after confirming that some of them had already been sent out of NCR before the seizure last week.According to officials, six injections from the batch were supplied to a dealer in Hyderabad. The dealer has been warned not to sell or use the stock. District drug controller Amandeep Chauhan said on Thursday that the department is now checking if more consignments were sent to other cities, including through e-commerce platforms.The case follows last week’s Rs 70-lakh seizure, in which 262 vials of suspected counterfeit Mounjaro injections were recovered during a raid near Super Mart-1. Two men, Mujammil Khan and supplier Avi Sharma, were arrested.“We are examining if supplies went beyond this batch and identifying all possible recipients. We have issued a notice to IndiaMart to share all details with us. They have requested some time to submit details,” Chauhan said.Authorities have sent samples for detailed laboratory testing, including sterility analysis, which requires a 14-day incubation period. Till the results are confirmed, circulation of the seized batch has been stopped.Chauhan said Sharma is now in five days’ judicial custody. Investigators said he operated from a flat in Gurgaon’s Sector 62, which was used solely for assembling and packaging the fake injections.Officials said the accused procured raw materials, including peptides and injection components, from China through an e-commerce platform. The products were then assembled at the flat, where investigators believe that online sources were used to imitate the original drug.According to the investigation, Sharma allegedly used 3D printing to create packaging cartons, labels, and instruction leaflets designed to resemble the genuine product. Officials said the peptides and injection pens were assembled into injectable units at the flat, where refrigeration facilities were used to store the materials.The counterfeit products were identified during inspection after several discrepancies were noticed. Officials found differences in shades of blue and red on the packaging compared with the original product. They also noted variations in typography and text alignment, while the illustration of the injection pen appeared darker and less sharp.“These visible discrepancies clearly indicated the product was not genuine,” Chauhan said.Officials also found that the injections were not stored at the required temperature range of 2°C to 8°C. The accused were unable to produce documents authorising the stocking or sale of the medicines.Investigators are now tracing both the source of the raw materials and the network of buyers. They are also examining possible links to Delhi’s Bhagirath Palace market to determine the full scale of the operation.With demand for tirzepatide-based medicines rising and prices remaining high, officials said such drugs are increasingly vulnerable to counterfeiting. The investigation is continuing.An Eli Lilly (India) spokesperson said, “We have been made aware of a recent development in relation to seizure of suspicious and counterfeit products that allegedly carry our product brand name Mounjaro (Tirzepatide). The said seizure resulted from an enforcement drive conducted in Gurgaon by officials from the state drugs regulatory authority. Lilly takes patient safety extremely seriously and welcomes the regulatory authority’s action against illicit medicines. We are actively supporting the investigation and will continue to work with regulatory and law enforcement authorities worldwide to protect patients from the risks of counterfeit products.”